- Metrology - Laboratory
- Metrology and Test Equipment
- Disintegration tester
- Pharma Test Apparatebau AG
Disintegration tester PTZ seriesautomatictabletfor the pharmaceutical industry





Add to favorites
Compare this product
Characteristics
- Test type
- disintegration
- Operating mode
- automatic
- Test material
- tablet
- Sector
- industrial, for the pharmaceutical industry
- Technology
- thermal
- Other characteristics
- stainless steel
Description
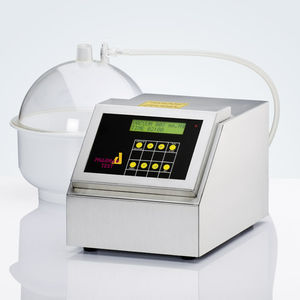
Tablet disintegration testing instruments are widely used in the pharmaceutical industry to evaluate the disintegration characteristics of formulations and the quality control of different dosage forms. All Pharma Test tablet and capsule disintegration testers are fully compliant with the current USP and EP Pharmacopeia and support both A-type and B-type baskets for regular or larger samples. They feature a stainless-steel housing, to be more GMP compliant than painted housing designs. Often these instruments are used in QC and IPC laboratories where most of the other equipment uses stainless steel as well. This way the Pharma Test instruments certainly do not look out of place.
The disintegration basket which holds the glass tubes and discs, is placed into a metal holder and held by a magnet. The instrument detects if a basket is inserted or not. The illuminated LC-display shows status information, like the elapsed testing time. The disintegration basket is moved up and down by 55mm (each way) at a rate of 30 times per minute. At test start it is moved smoothly into the testing medium.
The instrument’s heating system using a circulation pump warms the water inside the Plexiglas water bath up to the programmed target temperature, i.e. 37°C. A second sensor is used as a safety device which switches off the heater in case the temperature should rise outside the limit. A flow sensor, a thermal fuse and a thermal switch provide additional safety features to protect the system from overheating. The bath target temperature can be freely adjusted. Using the supplied temperature probe, the medium temperature can always be checked.
Catalogs
PTZ 100 and PTZ 300
8 Pages
Exhibitions
Meet this supplier at the following exhibition(s):

Other Pharma Test Apparatebau AG products
Galenic Instruments
Related Searches
- Measuring device
- Indentation hardness tester
- Benchtop indentation hardness tester
- Digital hardness tester
- Automatic test equipment
- Industrial testing system
- Portable indentation hardness tester
- Automatic testing system
- Manual test kit
- Tester with LCD screen
- Industrial test equipment
- Compact testing system
- Digital test equipment
- Diagnostic tester
- Laboratory test equipment
- Universal indentation hardness tester
- Tester with memory
- Compact test equipment
- Testing system with touchscreen
- R&D tester
*Prices are pre-tax. They exclude delivery charges and customs duties and do not include additional charges for installation or activation options. Prices are indicative only and may vary by country, with changes to the cost of raw materials and exchange rates.